- The ART Act 2021 sets the regulatory framework for all ART clinics and ART banks in India. It mandates the registration of a national registry for these institutions and imposes heavy regulations on donor gametes concerning informed consent and maintenance of medical records.
- Egg donation rules say donors have to be in the age bracket 23-35 years and should have one or more children. Plus, the donor can only donate to one couple. This is a ‘single donor’ rule that preserves genetic diversity.
- All gamete donations in India have to be done on a ‘no-profit’ basis. Hence, human gamete and human embryo trading is banned.
- Any child born through donor IVF is legally the child of the intended parents. Gamete donors do not get any parental rights.
- The Surrogacy Act 2021 only allows altruistic surrogacy, which means the surrogate mother has to be a close relative of the intended parents.
- Zemya IVF & Fertility Clinics strictly adheres to both the ART Act and the Surrogacy Act, ensuring that every patient is given care that is not only clinically excellent but also legally safeguarded.
India has been a hub for assisted reproductive technology services, and in this regard, the country’s legal framework for assisted reproductive technology services has seen significant and groundbreaking changes in recent years. The Assisted Reproductive Technology (Regulation) Act 2021 and the Surrogacy (Regulation) Act 2021 are two significant pieces of legislation that are probably the most comprehensive changes in the regulatory framework for fertility services in India’s history.
Being one of the dependable IVF Centers in Green Park, Zemya IVF & Fertility Clinics providing the best quality of IVF Treatment in Green Park, consider it a responsibility to comply strictly with the provisions of both Acts – in other words, each patient donor surrogate, and parent-to-be is guaranteed a service that is not only medically proficient but also legally and ethically one of the highest standard.
The ART Act 2021: An Overview
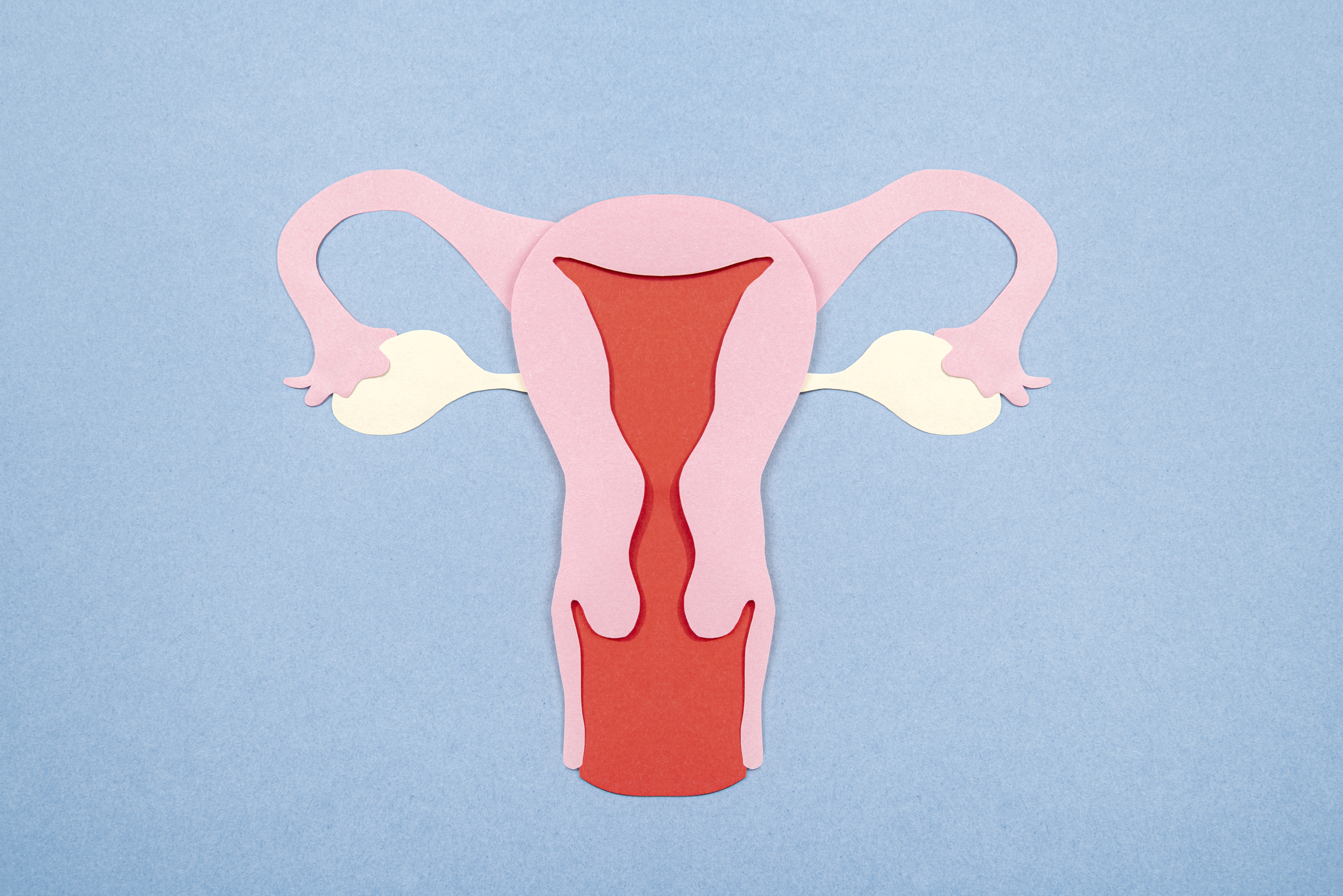
The Assisted Reproductive Technology (Regulation) Act 2021 was enacted by the central government to regulate ART Clinics and ART banks in India, to maintain a national registry of fertility clinics and donors, to safeguard the rights of donors, patients, and children conceived through ART procedures, and to prevent the misuse of ART. The Act shall apply to all ART Clinics providing fertility services, including IVF Treatment in Green Park, IUI Treatment in Green Park, ICSI Treatment in Green Park, Egg Freezing in Green Park, and ART banks for the collection, processing, and supply of human gametes and human embryos.
Every ART clinic and ART bank is required to be registered with the national assisted reproductive technology registry under the Act. Unregistered clinics cannot legally provide ART services, and sanctions for non-compliance include first-time fines and imprisonment for repeated offenses. The national registry established by the Act acts as a comprehensive record of all ART treatment undertaken in India, which it possible to trace.
Regulation of Gamete Donors Under the ART Act
One of the most important aspects of the ART Act is the detailed regulations regarding gamete donation, both egg donation and sperm donation, which have been included to ensure that the health and well-being of both the donor and the child conceived through gamete donation are protected.
Egg donors must be at least 23 years old and no more than 35 years old, and they should have given birth to a child at least once. This condition guarantees that oocyte donors comprehend the consequences of donation and have shown their ability to reproduce. One donor can only contribute eggs for one couple, a very strict rule that has been made not only to prevent extensive genetic spread from a single donor but also to minimize the risk of accidental consanguinity among children born from the same donor.
Donors are required to give written informed consent prior to all procedures in ART. There is a legal requirement for ART clinics to provide counseling to gamete donors prior to and subsequent to donation. There is a need for a complete medical record to be maintained by the ART clinic for all gamete donors, and these records are to be made available to the national registry.
The Act forbids trading in human gametes and human embryos. Therefore, all gamete donating in India has to be to the point of being selfless, with the donors only being paid for the actual medical costs and the reasonable inconvenience, not the gametes themselves. ART banks are the officially recognized main source for getting and providing donor gametes, and clinics must get donor sperm and donor eggs from the registered ART banks that are subject to the same conditions of supervision and regulation.
Blood group typing, genetic disease screening, and infectious disease testing of gamete donors are compulsory for all gamete donors before their gametes can be made available for use. The donors must also be tested for genetic diseases and chromosomal abnormalities that may affect the health of a child conceived through gametes. The full medical records of the donors must be maintained and disclosed to the intended parents.
Donor IVF: Legal Rights and Parental Rights
The most important part of the ART Act is unanimously the one concerning the legal rights of everyone involved in a donor IVF play. The law unambiguously states that a child born through donor IVF with donor egg or donor sperm is legally the biological child of the intended parents – the couple or single eligible individual who underwent the ART procedure – rather than the gamete donors. Donors don’t have any parental rights over a child born as a result of their donation, and similarly, a child born from the donation has no legal claim on the donor.
The intended parents are legally recognized as the parents of the child born as a result of the ART procedures from the moment of birth, and the legal parentage is protected, whether or not donor eggs, sperm, or embryos were used. Such a clear determination of the parental rights of the intended parents is one of the most significant protective measures set forth in the ART Act.
The law also provides for the execution of legal agreements between the ART clinic, the intended parents, and the ART bank prior to the initiation of any donor IVF cycle, thereby protecting the rights of all parties.
The Surrogacy (Regulation) Act 2021
Along with the ART Act, the Surrogacy (Regulation) Act 2021 is the legislation that regulates all surrogacy arrangements in India. It has completely replaced the previously unregulated situation with a well-defined and protective legal framework. The Act only allows altruistic surrogacythat is an altruistic surrogacy means a close relative of the intended parents becomes the surrogate mother and she does not receive any monetary compensation other than the medical expenses and the insurance coverage.
Arrangements where the surrogate mother is compensated for her services beyond what is documented as medical expenses are forbidden under the Act. This prohibition applies equally to both Indian nationals and foreign nationals, and same-sex couples and single individuals are excluded from availing of services in India, thus limiting access to heterosexual married Indian couples and widows and divorcees, following a ruling by the Supreme Court.
Who Is Eligible for Surrogacy Under the Act?
The Surrogacy Act outlines who can access surrogacy services. One of the things it says is that an Indian married couple would qualify for surrogacy only if they medically need it. In other words, it means the woman will not be able to conceive or carry the pregnancy to term due to the medical condition of her uterus, reproductive health, or the ability to carry a pregnancy as a whole. Such couples should have been married for at least five years. The age of the wife should be between 23 and 50, and the husband should be in the age range of 26 and 55.
The category of widows and divorcees between 35 and 45 years of age is also included under this provision, which was clarified with the aid of later developments. The category of same-sex couples continues to be excluded under the current law.
The Surrogate Mother: Legal and Medical Requirements
The surrogate mother must be a relative of the intended parents, and this makes India’s altruistic model distinct from other commercial models. The surrogate mother must be married and must have at least one biological child of her own. She must be aged between 25 and 35 years and may only act as a surrogate once in her lifetime. She must give her consent to all procedures involved in surrogacy in writing and must not be the egg provider in the same procedure, meaning that only gestational surrogacy is allowed.
The clinic that manages the surrogacy program will have to provide insurance for the surrogate mother to cover all her medical expenses arising from the pregnancy, delivery, and any complications resulting from the procedures involved in surrogacy. The insurance will have to be in place prior to the embryo transfer.
Registration, Compliance, and Grievance Redressal
In addition, all ART clinics offering surrogacy facilities must be separately registered under the Surrogacy Act. The Act provides for the establishment of national and state-level surrogacy boards, which shall be responsible for overseeing the implementation of the policies. In addition, a grievance redressal cell must be set up at the national as well as the state government levels, which shall redress the grievances of intended parents, surrogates, and donors.
The national registries set up under both the Acts shall maintain records of all ART procedures, donor cycles, surrogacy, as well as children born as a result of these procedures. Such registries shall safeguard the interests of all parties involved, including the rights of the children born as a result of donor insemination to have access to non-identifying medical information.
In Zemya IVF & Fertility Clinics, full registration, compliance documents, legal agreements, consent, and insurance are stringently followed for all patients seeking surrogacy or IVF with donors, thus making this clinic one of the most compliant and ethical clinics in South Delhi.